| バックグラウンド: | The Von Hippel-Lindau (VHL) protein is a substrate recognition component of an E3 ubiquitin ligase complex containing elongin BC (TCEB1 and TCEB2), cullin 1 (CUL1), and RING-box protein 1 (RBX1). VHL protein has been shown to exist as three distinct isoforms resulting from alternatively spliced transcript variants. Loss of VHL protein function results in a dominantly inherited familial cancer syndrome that manifests as angiomas of the retina, hemangioblastomas of the central nervous system, renal clear-cell carcinomas and pheochromocytomas. Under normoxic conditions, VHL directs the ubiquitylation and subsequent proteosomal degradation of the hypoxia inducible factor HIF alpha, maintaining very low levels of HIF α in the cell. Cellular exposure to hypoxic conditions, or loss of VHL protein function, results in increased HIF α protein levels and increased expression of HIF-induced gene products, many of which are angiogenesis factors such as vascular endothelial growth factor (VEGF). Thus, loss of VHL protein function is believed to contribute to the formation of highly vascular neoplasias. In addition to HIF alpha, VHL is known to regulate the ubiquitylation of several other proteins, including tat-binding protein 1 (TBP-1), the atypical protein kinase C lambda (aPKCµ, and two subunits of the multiprotein RNA Polymerase II complex (RPB1 and RPB7). Interactions with elongin BC, RPB1, RPB7 and the pVHL-associated KRAB-A domain containing protein (VHLaK) suggest that VHL may also play a more direct role in transcriptional repression. |
| シノニム: | Von Hippel-Lindau disease tumor suppressor; pVHL; Protein G7; VHL |
| UniProt Protein Function: | VHL: Involved in the ubiquitination and subsequent proteasomal degradation via the von Hippel-Lindau ubiquitination complex. Seems to act as target recruitment subunit in the E3 ubiquitin ligase complex and recruits hydroxylated hypoxia- inducible factor (HIF) under normoxic conditions. Involved in transcriptional repression through interaction with HIF1A, HIF1AN and histone deacetylases. Ubiquitinates, in an oxygen-responsive manner, ADRB2. Component of the VCB (VHL-Elongin BC-CUL2) complex; this complex acts as a ubiquitin-ligase E3 and directs proteasome- dependent degradation of targeted proteins. Interacts with CUL2; this interaction is dependent on the integrity of the trimeric VBC complex. Interacts (via the beta domain) with HIF1A (via the NTAD domain); this interaction mediates degradation of HIF1A in normoxia and, in hypoxia, prevents ubiqitination and degradation of HIF1A by mediating hypoxia-induced translocation to the nucleus, a process which requires a hypoxia-dependent regulatory signal. Interacts with ADRB2; the interaction, in normoxia, is dependent on hydroxylation of ADRB2 and the subsequent VCB- mediated ubiquitination and degradation of ADRB2. Under hypoxia, hydroxylation, interaction with VHL, ubiquitination and subsequent degradation of ADRB2 are dramatically decreased. Interacts with RNF139, USP33 and PHF17. Found in a complex composed of LIMD1, VHL, EGLN1/PHD2, TCEB2 AND CUL2. Isoform 1 and isoform 3 interact with LIMD1 (via LIM zinc-binding 2), AJUBA (via LIM domains) and WTIP (via LIM domains). Interacts with EPAS1. Expressed in the adult and fetal brain and kidney. 3 isoforms of the human protein are produced by alternative splicing. |
| UniProt Protein Details: | Protein type:Ubiquitin ligase; Tumor suppressor Chromosomal Location of Human Ortholog: 3p25.3 Cellular Component: nucleoplasm; intermediate filament cytoskeleton; mitochondrion; membrane; endoplasmic reticulum; cytosol; nucleus Molecular Function:protein binding; enzyme binding; ubiquitin-protein ligase activity; transcription factor binding Biological Process: negative regulation of cell proliferation; regulation of transcription, DNA-dependent; protein stabilization; positive regulation of transcription, DNA-dependent; cell morphogenesis; protein ubiquitination; negative regulation of transcription from RNA polymerase II promoter; proteolysis; positive regulation of cell differentiation; negative regulation of apoptosis Disease: Erythrocytosis, Familial, 2; Von Hippel-lindau Syndrome; Renal Cell Carcinoma, Nonpapillary; Pheochromocytoma |
| NCBI Summary: | Von Hippel-Lindau syndrome (VHL) is a dominantly inherited familial cancer syndrome predisposing to a variety of malignant and benign tumors. A germline mutation of this gene is the basis of familial inheritance of VHL syndrome. The protein encoded by this gene is a component of the protein complex that includes elongin B, elongin C, and cullin-2, and possesses ubiquitin ligase E3 activity. This protein is involved in the ubiquitination and degradation of hypoxia-inducible-factor (HIF), which is a transcription factor that plays a central role in the regulation of gene expression by oxygen. RNA polymerase II subunit POLR2G/RPB7 is also reported to be a target of this protein. Alternatively spliced transcript variants encoding distinct isoforms have been observed. [provided by RefSeq, Jul 2008] |
| UniProt Code: | P40337 |
| NCBI GenInfo Identifier: | 4033778 |
| NCBI Gene ID: | 7428 |
| NCBI Accession: | P40337.2 |
| UniProt Secondary Accession: | P40337,Q13599, Q6PDA9, B2RE45, |
| UniProt Related Accession: | P40337 |
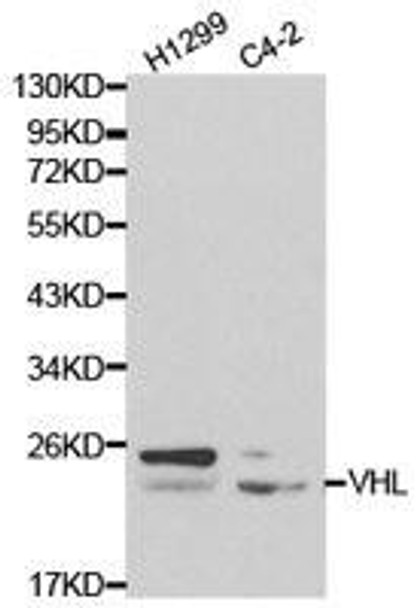
| Molecular Weight: | Calculated: 18kDa/19kDa/24kDaObserved: 24kDa |
| NCBI Full Name: | Von Hippel-Lindau disease tumor suppressor |
| NCBI Synonym Full Names: | von Hippel-Lindau tumor suppressor, E3 ubiquitin protein ligase |
| NCBI Official Symbol: | VHL |
| NCBI Official Synonym Symbols: | RCA1; VHL1; pVHL; HRCA1 |
| NCBI Protein Information: | von Hippel-Lindau disease tumor suppressor; protein G7; elongin binding protein |
| UniProt Protein Name: | Von Hippel-Lindau disease tumor suppressor |
| UniProt Synonym Protein Names: | Protein G7; pVHL |
| Protein Family: | Von Hippel-Lindau disease tumor suppressor |
| UniProt Gene Name: | VHL |
| UniProt Entry Name: | VHL_HUMAN |